What Will the Concentration of Cd2+ at the Moment Before Fe2+ Begins to Precipitate?
Chapter 15. Equilibria of Other Reaction Classes
15.1 Precipitation and Dissolution
Learning Objectives
Past the end of this section, you lot will be able to:
- Write chemic equations and equilibrium expressions representing solubility equilibria
- Carry out equilibrium computations involving solubility, equilibrium expressions, and solute concentrations
The preservation of medical laboratory blood samples, mining of ocean water for magnesium, conception of over-the-counter medicines such as Milk of Magnesia and antacids, and treating the presence of difficult water in your dwelling house'south water supply are merely a few of the many tasks that involve controlling the equilibrium between a slightly soluble ionic solid and an aqueous solution of its ions.
In some cases, nosotros want to prevent dissolution from occurring. Molar decay, for example, occurs when the calcium hydroxylapatite, which has the formula Cafive(PO4)3(OH), in our teeth dissolves. The dissolution process is aided when bacteria in our mouths banquet on the sugars in our diets to produce lactic acid, which reacts with the hydroxide ions in the calcium hydroxylapatite. Preventing the dissolution prevents the decay. On the other manus, sometimes we want a substance to deliquesce. Nosotros want the calcium carbonate in a chewable antacid to dissolve because the [latex]\text{CO}_3^{\;\;2-}[/latex] ions produced in this process assistance soothe an upset stomach.
In this section, we will find out how we can control the dissolution of a slightly soluble ionic solid by the application of Le Châtelier'due south principle. We will also acquire how to use the equilibrium constant of the reaction to determine the concentration of ions present in a solution.
The Solubility Product Constant
Silver chloride is what's known as a sparingly soluble ionic solid (Figure 1). Call back from the solubility rules in an earlier chapter that halides of Ag+ are not normally soluble. Even so, when nosotros add together an excess of solid AgCl to water, it dissolves to a small extent and produces a mixture consisting of a very dilute solution of Ag+ and Cl– ions in equilibrium with undissolved silver chloride:
[latex]\text{AgCl}(due south)\;\underset{\text{precipitation}}{\overset{\text{dissolution}}{\rightleftharpoons}}\;\text{Ag}^{+}(aq)\;+\;\text{Cl}^{-}(aq)[/latex]
This equilibrium, like other equilibria, is dynamic; some of the solid AgCl continues to deliquesce, but at the same time, Ag+ and Cl– ions in the solution combine to produce an equal amount of the solid. At equilibrium, the opposing processes take equal rates.
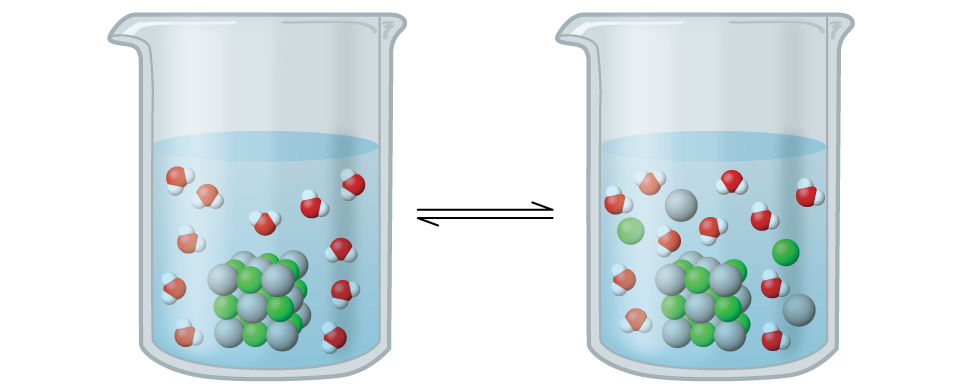
The equilibrium constant for the equilibrium between a slightly soluble ionic solid and a solution of its ions is called the solubility product (Grand sp) of the solid. Remember from the chapter on solutions and colloids that we use an ion'southward concentration as an approximation of its activity in a dilute solution. For silvery chloride, at equilibrium:
[latex]\text{AgCl}(southward)\;{\rightleftharpoons}\;\text{Ag}^{+}(aq)\;+\;\text{Cl}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Ag}^{+}(aq)][\text{Cl}^{-}(aq)][/latex]
When looking at dissolution reactions such every bit this, the solid is listed as a reactant, whereas the ions are listed as products. The solubility product constant, as with every equilibrium constant expression, is written as the product of the concentrations of each of the ions, raised to the ability of their stoichiometric coefficients. Here, the solubility product constant is equal to Ag+ and Cl– when a solution of silver chloride is in equilibrium with undissolved AgCl. In that location is no denominator representing the reactants in this equilibrium expression since the reactant is a pure solid; therefore [AgCl] does not appear in the expression for One thousand sp.
Some common solubility products are listed in Tabular array 1 according to their K sp values, whereas a more extensive compilation of products appears in Appendix J. Each of these equilibrium constants is much smaller than 1 considering the compounds listed are only slightly soluble. A small M sp represents a system in which the equilibrium lies to the left, so that relatively few hydrated ions would be present in a saturated solution.
| Substance | K sp at 25 °C |
|---|---|
| CuCl | 1.ii × 10–6 |
| CuBr | six.27 × 10–9 |
| AgI | i.5 × 10–16 |
| PbS | 7 × 10–29 |
| Al(OH)three | ii × 10–32 |
| Iron(OH)3 | iv × x–38 |
| Table 1. Common Solubility Products by Decreasing Equilibrium Constants | |
Case i
Writing Equations and Solubility Products
Write the ionic equation for the dissolution and the solubility product expression for each of the post-obit slightly soluble ionic compounds:
(a) AgI, silver iodide, a solid with antiseptic properties
(b) CaCO3, calcium carbonate, the active ingredient in many over-the-counter chewable antacids
(c) Mg(OH)ii, magnesium hydroxide, the agile ingredient in Milk of Magnesia
(d) Mg(NH4)PO4, magnesium ammonium phosphate, an essentially insoluble substance used in tests for magnesium
(e) Ca5(PO4)3OH, the mineral apatite, a source of phosphate for fertilizers
(Hint: When determining how to break (d) and (east) upwardly into ions, refer to the list of polyatomic ions in the department on chemic nomenclature.)
Solution
(a) [latex]\text{AgI}(southward)\;{\rightleftharpoons}\;\text{Ag}^{+}(aq)\;+\;\text{I}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Ag}^{+}][\text{I}^{-}][/latex]
(b) [latex]\text{CaCO}_3(s)\;{\rightleftharpoons}\;\text{Ca}^{ii+}(aq)\;+\;\text{CO}_3^{\;\;two-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Ca}^{2+}][\text{CO}_3^{\;\;two-}][/latex]
(c) [latex]\text{Mg(OH)}_2(s)\;{\rightleftharpoons}\;\text{Mg}^{ii+}(aq)\;+\;2\text{OH}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Mg}^{2+}][\text{OH}^{-}]^2[/latex]
(d) [latex]\text{Mg(NH}_4)\text{PO}_4(south)\;{\rightleftharpoons}\;\text{Mg}^{two+}(aq)\;+\;\text{NH}_4^{\;\;+}(aq)\;+\;\text{PO}_4^{\;\;iii-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Mg}^{2+}][\text{NH}_4^{\;\;+}][\text{PO}_4^{\;\;3-}][/latex]
(e) [latex]\text{Ca}_5(\text{PO}_4)iii\text{OH}(s)\;{\rightleftharpoons}\;5\text{Ca}^{2+}(aq)\;+\;iii\text{PO}_4^{\;\;3-}(aq)\;+\;\text{OH}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Ca}^{2+}]^5[\text{PO}_4^{\;\;3-}]^3[\text{OH}^{-}][/latex]
Cheque Your Learning
Write the ionic equation for the dissolution and the solubility product for each of the following slightly soluble compounds:
(a) BaSO4
(b) AgtwoSo4
(c) Al(OH)3
(d) Pb(OH)Cl
Reply:
(a) [latex]\text{BaSO}_4(south)\;{\rightleftharpoons}\;\text{Ba}^{ii+}(aq)\;+\;\text{And so}_4^{\;\;2-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Ba}^{2+}][\text{SO}_4^{\;\;2-}][/latex]; (b) [latex]\text{Ag}_2\text{And then}_4(due south)\;{\rightleftharpoons}\;2\text{Ag}^{+}(aq)\;+\;\text{SO}_4^{\;\;2-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Ag}^{+}]^2[\text{And then}_4^{\;\;2-}][/latex]; (c) [latex]\text{Al(OH)}_3(s)\;{\rightleftharpoons}\;\text{Al}^{2+}(aq)\;+\;iii\text{OH}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Al}^{3+}][\text{OH}^{-}]^3[/latex]; (d) [latex]\text{Pb(OH)Cl}(south)\;{\rightleftharpoons}\;\text{Pb}^{two+}(aq)\;+\;\text{OH}^{-}(aq)\;+\;\text{Cl}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Atomic number 82}^{2+}][\text{OH}^{-}][\text{Cl}^{-}][/latex]
Now we will extend the discussion of Yard sp and show how the solubility product constant is determined from the solubility of its ions, likewise as how K sp can be used to determine the molar solubility of a substance.
K sp and Solubility
Recall that the definition of solubility is the maximum possible concentration of a solute in a solution at a given temperature and pressure. We tin determine the solubility product of a slightly soluble solid from that mensurate of its solubility at a given temperature and force per unit area, provided that the but meaning reaction that occurs when the solid dissolves is its dissociation into solvated ions, that is, the merely equilibrium involved is:
[latex]\text{M}_p\text{X}_q(s)\;{\rightleftharpoons}\;p\text{One thousand}^{\text{thou+}}(aq)\;+\;q\text{X}^{\text{due north-}}(aq)[/latex]
In this case, nosotros calculate the solubility product by taking the solid'south solubility expressed in units of moles per liter (mol/Fifty), known as its molar solubility.
Example two
Calculation of K sp from Equilibrium Concentrations
Nosotros began the chapter with an breezy discussion of how the mineral fluorite (Introduction to Affiliate fifteen) is formed. Fluorite, CaF2, is a slightly soluble solid that dissolves according to the equation:
[latex]\text{CaF}_2(s)\;{\rightleftharpoons}\;\text{Ca}^{2+}(aq)\;+\;two\text{F}^{-}(aq)[/latex]
The concentration of Ca2+ in a saturated solution of CaFii is 2.fifteen × 10–4 M; therefore, that of F– is 4.thirty × 10–4 Yard, that is, twice the concentration of Ca2+. What is the solubility product of fluorite?
Solution
Kickoff, write out the One thousand sp expression, so substitute in concentrations and solve for K sp:
[latex]\text{CaF}_2(south)\;{\rightleftharpoons}\;\text{Ca}^{2+}(aq)\;+\;2\text{F}^{-}(aq)[/latex]
A saturated solution is a solution at equilibrium with the solid. Thus:
[latex]K_{\text{sp}} = [\text{Ca}^{two+}][\text{F}^{-}]^2 = (2.1\;\times\;10^{-four})(four.ii\;\times\;x^{-4})^2 = 3.vii\;\times\;10^{-eleven}[/latex]
Every bit with other equilibrium constants, nosotros practise not include units with K sp.
Check Your Learning
In a saturated solution that is in contact with solid Mg(OH)2, the concentration of Mg2+ is 1.31 × 10–4 One thousand. What is the solubility product for Mg(OH)2?
[latex]\text{Mg(OH)}_2(southward)\;{\rightleftharpoons}\;\text{Mg}^{ii+}(aq)\;+\;two\text{OH}^{-}(aq)[/latex]
Case 3
Determination of Tooth Solubility from K sp
The Grand sp of copper(I) bromide, CuBr, is half-dozen.3 × ten–9. Calculate the molar solubility of copper bromide.
Solution
The solubility product constant of copper(I) bromide is 6.three × x–nine.
The reaction is:
[latex]\text{CuBr}(southward)\;{\rightleftharpoons}\;\text{Cu}^{+}(aq)\;+\;\text{Br}^{-}(aq)[/latex]
Beginning, write out the solubility production equilibrium constant expression:
[latex]K_{\text{sp}} = [\text{Cu}^{+}][\text{Br}^{-}][/latex]
Create an ICE tabular array (equally introduced in the chapter on key equilibrium concepts), leaving the CuBr column empty as it is a solid and does not contribute to the K sp:
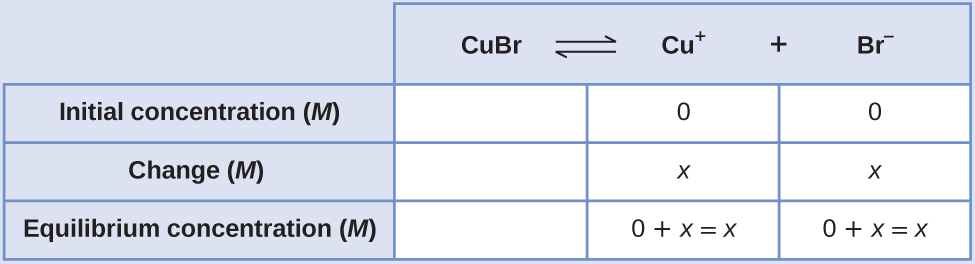
At equilibrium:
[latex]K_{\text{sp}} = [\text{Cu}^{+}][\text{Br}^{-}][/latex]
[latex]six.3\;\times\;10^{-9} = (x)(10) = x^2[/latex]
[latex]x = \sqrt{(six.three\;\times\;10^{-nine})} = 7.ix\;\times\;10^{-5}[/latex]
Therefore, the tooth solubility of CuBr is 7.9 × ten–5 M.
Check Your Learning
The K sp of AgI is i.5 × x–xvi. Calculate the tooth solubility of argent iodide.
Example 4
Determination of Molar Solubility from K sp, Function Two
The G sp of calcium hydroxide, Ca(OH)2, is i.three × 10–6. Calculate the molar solubility of calcium hydroxide.
Solution
The solubility product constant of calcium hydroxide is i.three × ten–vi.
The reaction is:
[latex]\text{Ca(OH)}_2(s)\;{\rightleftharpoons}\;\text{Ca}^{ii+}(aq)\;+\;ii\text{OH}^{-}(aq)[/latex]
Offset, write out the solubility product equilibrium constant expression:
[latex]K_{\text{sp}} = [\text{Ca}^{two+}][\text{OH}^{-}]^2[/latex]
Create an Water ice table, leaving the Ca(OH)ii column empty as information technology is a solid and does not contribute to the K sp:
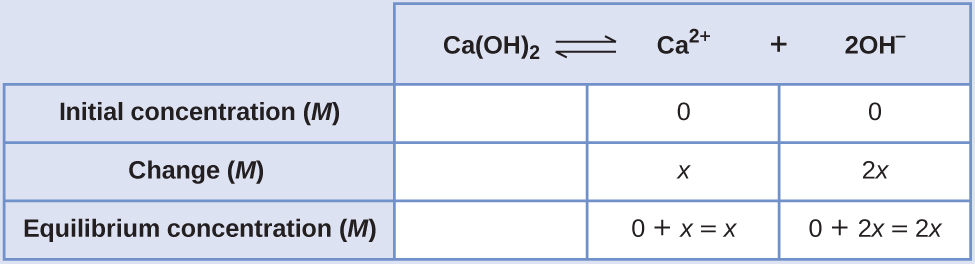
At equilibrium:
[latex]K_{\text{sp}} = [\text{Ca}^{2+}][\text{OH}^{-}]^2[/latex]
[latex]1.three\;\times\;x^{-6} = (ten)(2x)^2 = (x)(4x^2) = 4x^3[/latex]
[latex]x = \sqrt[3]{\frac{ane.3\;\times\;10^{-6}}{4}} = 6.9\;\times\;10^{-iii}[/latex]
Therefore, the molar solubility of Ca(OH)2 is i.3 × ten–2 M.
Check Your Learning
The K sp of PbI2 is 1.4 × 10–8. Calculate the molar solubility of lead(Ii) iodide.
Annotation that solubility is non always given every bit a tooth value. When the solubility of a compound is given in some unit of measurement other than moles per liter, nosotros must convert the solubility into moles per liter (i.e., molarity) in order to use it in the solubility product constant expression. Case 5 shows how to perform those unit of measurement conversions before determining the solubility product equilibrium.
Example 5
Determination of K sp from Gram Solubility
Many of the pigments used by artists in oil-based paints (Figure ii) are sparingly soluble in water. For instance, the solubility of the creative person's pigment chrome yellow, PbCrOfour, is four.6 × ten–6 chiliad/L. Determine the solubility product equilibrium constant for PbCrO4.

Solution
We are given the solubility of PbCrO4 in grams per liter. If nosotros convert this solubility into moles per liter, we can find the equilibrium concentrations of Atomic number 82ii+ and [latex]\text{CrO}_4^{\;\;2-}[/latex], so 1000 sp:
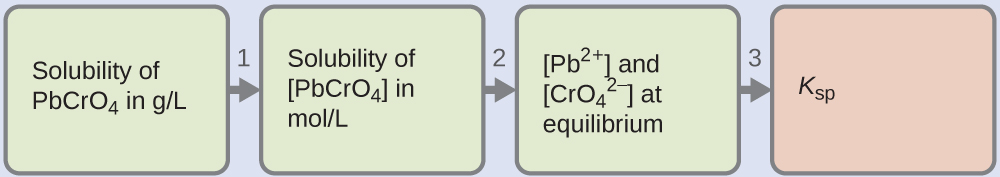
- Utilize the molar mass of PbCrO4 [latex](\frac{323.ii\;\text{m}}{1\;\text{mol}})[/latex] to convert the solubility of PbCrO4 in grams per liter into moles per liter:
[latex][\text{PbCrO}_4] = \frac{4.6\;\times\;10^{-half dozen}\;\text{1000}\;\text{PbCrO}_4}{1\;\text{L}}\;\times\;\frac{1\;\text{mol}\;\text{PbCrO}_4}{323.2\;\text{yard\;PbCrO}_4}[/latex]
[latex]= \frac{i.iv\;\times\;10^{-8}\;\text{mol\;PbCrO}_4}{one\;\text{50}}[/latex]
[latex]= 1.4\;\times\;x^{-viii}\;M[/latex] - The chemic equation for the dissolution indicates that one mol of PbCrO4 gives 1 mol of Pb2+(aq) and i mol of [latex]\text{CrO}_4^{\;\;two-}(aq)[/latex]:
[latex]\text{PbCrO}_4(s)\;{\rightleftharpoons}\;\text{Pb}^{2+}(aq)\;+\;\text{CrO}_4^{\;\;2-}(aq)[/latex]
Thus, both [Lead2+] and [latex][\text{CrO}_4^{\;\;ii-}][/latex] are equal to the tooth solubility of PbCrO4:
[latex][\text{Pb}^{ii+}] = [\text{CrO}_4^{\;\;2-}] = one.four\;\times\;10^{-eight}\;M[/latex]
- Solve. Thou sp = [Lead2+][latex][\text{CrO}_4^{\;\;2-}][/latex] = (1.4 × 10–8)(1.4 × x–8) = 2.0 × x–xvi
Bank check Your Learning
The solubility of TlCl [thallium(I) chloride], an intermediate formed when thallium is beingness isolated from ores, is three.46 grams per liter at 20 °C. What is its solubility product?
Example 6
Calculating the Solubility of HgtwoCl2
Calomel, HgiiCltwo, is a compound equanimous of the diatomic ion of mercury(I), [latex]\text{Hg}_2^{\;\;2+}[/latex], and chloride ions, Cl–. Although most mercury compounds are at present known to be poisonous, eighteenth-century physicians used calomel as a medication. Their patients rarely suffered any mercury poisoning from the treatments because calomel is quite insoluble:
[latex]\text{Hg}_2\text{Cl}_2(s)\;{\rightleftharpoons}\;\text{Hg}_2^{\;\;2+}(aq)\;+\;ii\text{Cl}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = 1.1\;\times\;10^{-eighteen}[/latex]
Calculate the molar solubility of Hg2Clii.
Solution
The molar solubility of Hg2Cl2 is equal to the concentration of [latex]\text{Hg}_2^{\;\;2+}[/latex] ions because for each 1 mol of Hg2Cl2 that dissolves, 1 mol of [latex]\text{Hg}_2^{\;\;2+}[/latex] forms:
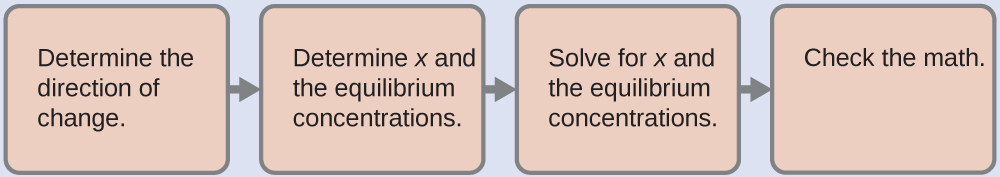
- Determine the direction of change. Earlier any Hg2Cl2 dissolves, Q is aught, and the reaction will shift to the right to accomplish equilibrium.
- Make up one's mind ten and equilibrium concentrations. Concentrations and changes are given in the following Water ice tabular array:
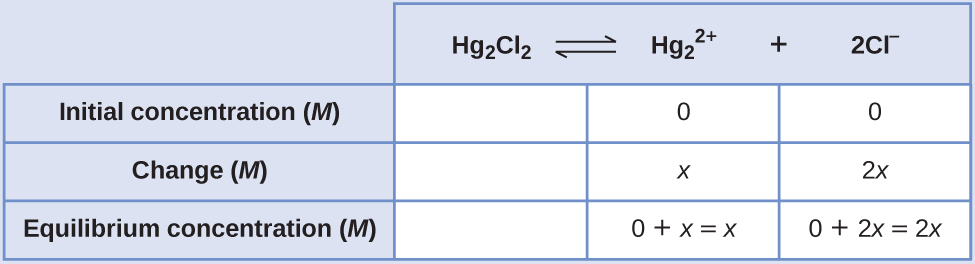
Annotation that the modify in the concentration of Cl– (twox) is twice as large as the change in the concentration of [latex]\text{Hg}_2^{\;\;2+}[/latex] (x) because 2 mol of Cl– forms for each one mol of [latex]\text{Hg}_2^{\;\;2+}[/latex] that forms. Hg2Clii is a pure solid, and then information technology does not announced in the calculation.
- Solve for x and the equilibrium concentrations. Nosotros substitute the equilibrium concentrations into the expression for Thou sp and calculate the value of x:
[latex]K_{\text{sp}} = [\text{Hg}_2^{\;\;2+}][\text{Cl}^{-}]^2[/latex]
[latex]1.1\;\times\;10^{-xviii} = (ten)(2x)^ii[/latex]
[latex]4x^iii = ane.one\;\times\;10^{-18}[/latex]
[latex]10 = \sqrt[3]{(\frac{i.ane\;\times\;10^{-18}}{iv})} = 6.v\;\times\;10^{-7}\;M[/latex]
[latex][\text{Hg}_2^{\;\;2+}] = half-dozen.5\;\times\;10^{-7}\;M = six.5\;\times\;ten^{-7}\;Chiliad[/latex]
[latex][\text{Cl}^{-}] = 2x = 2(6.5\;\times\;10^{-7}) = 1.3\;\times\;10^{-half-dozen}\;Thou[/latex]
The molar solubility of Hg2Cl2 is equal to [latex][\text{Hg}_2^{\;\;two+}][/latex], or vi.five × 10–vii M.
- Check the work. At equilibrium, Q = K sp:
[latex]Q = [\text{Hg}_2^{\;\;2+}][\text{Cl}^{-}]^2 = (6.5\;\times\;10^{-7})(1.three\;\times\;x^{-6})^2 = 1.1\;\times\;10^{-18}[/latex]
The calculations check.
Cheque Your Learning
Make up one's mind the tooth solubility of MgF2 from its solubility product: Grand sp = 6.four × 10–ix.
Tabulated 1000 sp values can besides be compared to reaction quotients calculated from experimental data to tell whether a solid volition precipitate in a reaction under specific conditions: Q equals K sp at equilibrium; if Q is less than K sp, the solid will dissolve until Q equals M sp; if Q is greater than G sp, precipitation will occur at a given temperature until Q equals K sp.
Using Barium Sulfate for Medical Imaging
Various types of medical imaging techniques are used to aid diagnoses of illnesses in a noninvasive manner. One such technique utilizes the ingestion of a barium compound earlier taking an X-ray epitome. A suspension of barium sulfate, a chalky powder, is ingested by the patient. Since the Thou sp of barium sulfate is 1.i × 10–10, very piddling of it dissolves as it coats the lining of the patient's abdominal tract. Barium-coated areas of the digestive tract so appear on an Ten-ray as white, assuasive for greater visual detail than a traditional X-ray (Figure 3).
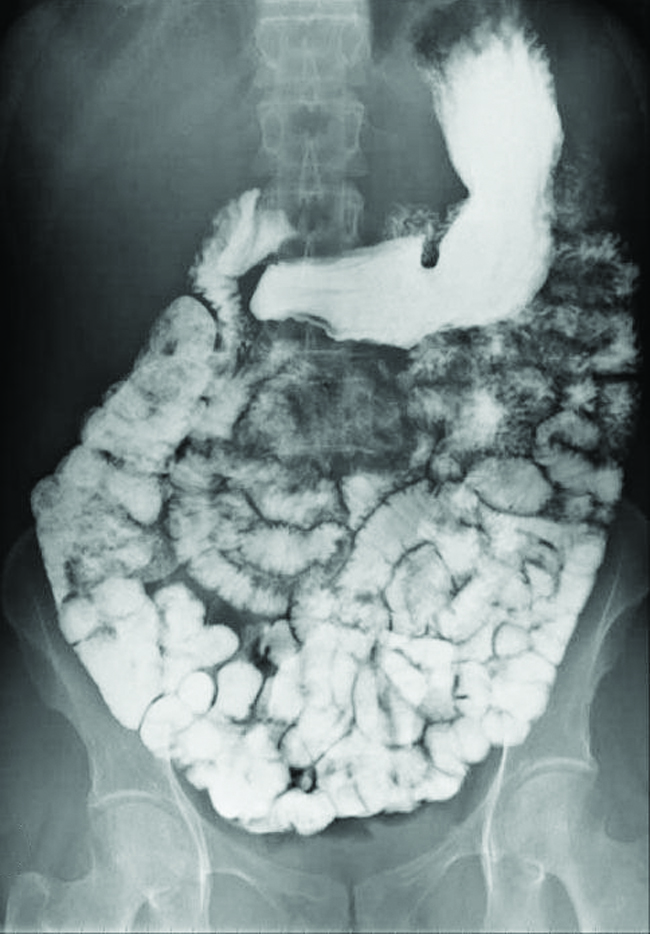
Farther diagnostic testing can be done using barium sulfate and fluoroscopy. In fluoroscopy, a continuous X-ray is passed through the body so the doctor can monitor, on a TV or computer screen, the barium sulfate's movement equally it passes through the digestive tract. Medical imaging using barium sulfate can be used to diagnose acrid reflux disease, Crohn'south disease, and ulcers in addition to other conditions.
Visit this website for more information on how barium is used in medical diagnoses and which atmospheric condition it is used to diagnose.
Predicting Precipitation
The equation that describes the equilibrium between solid calcium carbonate and its solvated ions is:
[latex]\text{CaCO}_3(s)\;{\rightleftharpoons}\;\text{Ca}^{2+}(aq)\;+\;\text{CO}_3^{\;\;2-}(aq)[/latex]
We can establish this equilibrium either by calculation solid calcium carbonate to water or by mixing a solution that contains calcium ions with a solution that contains carbonate ions. If we add calcium carbonate to water, the solid will deliquesce until the concentrations are such that the value of the reaction quotient [latex](\text{Q} = [\text{Ca}^{2+}][\text{CO}_3^{\;\;2-}])[/latex] is equal to the solubility product (K sp = 8.7 × x–9). If we mix a solution of calcium nitrate, which contains Ca2+ ions, with a solution of sodium carbonate, which contains [latex]\text{CO}_3^{\;\;2-}[/latex] ions, the slightly soluble ionic solid CaCOthree volition precipitate, provided that the concentrations of Caii+ and [latex]\text{CO}_3^{\;\;2-}[/latex] ions are such that Q is greater than K sp for the mixture. The reaction shifts to the left and the concentrations of the ions are reduced by germination of the solid until the value of Q equals Grand sp. A saturated solution in equilibrium with the undissolved solid will outcome. If the concentrations are such that Q is less than K sp, and so the solution is non saturated and no precipitate will form.
We can compare numerical values of Q with K sp to predict whether precipitation will occur, equally Case seven shows. (Annotation: Since all forms of equilibrium constants are temperature dependent, we will assume a room temperature environment going forward in this chapter unless a unlike temperature value is explicitly specified.)
Instance 7
Precipitation of Mg(OH)2
The first step in the preparation of magnesium metallic is the atmospheric precipitation of Mg(OH)2 from ocean water by the addition of lime, Ca(OH)2, a readily bachelor cheap source of OH– ion:
[latex]\text{Mg(OH)}_2(s)\;{\rightleftharpoons}\;\text{Mg}^{2+}(aq)\;+\;2\text{OH}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = 8.9\;\times\;10^{-12}[/latex]
The concentration of Mg2+(aq) in bounding main water is 0.0537 One thousand. Will Mg(OH)ii precipitate when enough Ca(OH)2 is added to give a [OH–] of 0.0010 Thou?
Solution
This problem asks whether the reaction:
[latex]\text{Mg(OH)}_2(s)\;{\leftrightharpoons}\;\text{Mg}^{ii+}(aq)\;+\;two\text{OH}^{-}(aq)[/latex]
shifts to the left and forms solid Mg(OH)two when [Mg2+] = 0.0537 M and [OH–] = 0.0010 M. The reaction shifts to the left if Q is greater than K sp. Adding of the reaction quotient under these conditions is shown here:
[latex]Q = [\text{Mg}^{two+}][\text{OH}^{-}]^ii = (0.0537)(0.0010)^two = five.4\;\times\;10^{-viii}[/latex]
Because Q is greater than K sp (Q = 5.4 × 10–viii is larger than K sp = viii.9 × 10–12), nosotros tin expect the reaction to shift to the left and form solid magnesium hydroxide. Mg(OH)2(s) forms until the concentrations of magnesium ion and hydroxide ion are reduced sufficiently so that the value of Q is equal to One thousand sp.
Check Your Learning
Utilize the solubility product in Appendix J to determine whether CaHPO4 volition precipitate from a solution with [Catwo+] = 0.0001 One thousand and [latex][\text{HPO}_4^{\;\;two-}][/latex] = 0.001 M.
Respond:
No precipitation of CaHPO4; Q = one × 10–vii, which is less than One thousand sp
Example 8
Precipitation of AgCl upon Mixing Solutions
Does silver chloride precipitate when equal volumes of a ii.0 × ten–4–M solution of AgNO3 and a 2.0 × ten–4–G solution of NaCl are mixed?
(Note: The solution likewise contains Na+ and [latex]\text{NO}_3^{\;\;-}[/latex] ions, merely when referring to solubility rules, one can run into that sodium nitrate is very soluble and cannot form a precipitate.)
Solution
The equation for the equilibrium between solid silvery chloride, silver ion, and chloride ion is:
[latex]\text{AgCl}(s)\;{\rightleftharpoons}\;\text{Ag}^{+}(aq)\;+\;\text{Cl}^{-}(aq)[/latex]
The solubility production is i.6 × x–10 (see Appendix J).
AgCl will precipitate if the reaction caliber calculated from the concentrations in the mixture of AgNOiii and NaCl is greater than K sp. The book doubles when we mix equal volumes of AgNO3 and NaCl solutions, so each concentration is reduced to half its initial value. Consequently, immediately upon mixing, [Ag+] and [Cl–] are both equal to:
[latex]\frac{1}{2}(2.0\;\times\;10^{-four})\;Thou = 1.0\;\times\;10^{-four}\;Thou[/latex]
The reaction quotient, Q, is momentarily greater than One thousand sp for AgCl, so a supersaturated solution is formed:
[latex]Q = [\text{Ag}^{+}][\text{Cl}^{-}] = (ane.0\;\times\;x^{-4})(1.0\;\times\;x^{-4}) = i.0\;\times\;10^{-eight}\;{\textgreater}\;K_{\text{sp}}[/latex]
Since supersaturated solutions are unstable, AgCl will precipitate from the mixture until the solution returns to equilibrium, with Q equal to Grand sp.
Check Your Learning
Will KClO4 precipitate when twenty mL of a 0.050-Thousand solution of K+ is added to 80 mL of a 0.l-M solution of [latex]\text{ClO}_4^{\;\;-}[/latex]? (Call back to calculate the new concentration of each ion after mixing the solutions before plugging into the reaction quotient expression.)
Answer:
No, Q = 4.0 × 10–three, which is less than K sp = 1.05 × x–2
In the previous two examples, nosotros have seen that Mg(OH)ii or AgCl precipitate when Q is greater than K sp. In general, when a solution of a soluble salt of the Mm+ ion is mixed with a solution of a soluble salt of the 10n– ion, the solid, Thou p X q precipitates if the value of Q for the mixture of Mm+ and Xn– is greater than K sp for Chiliad p X q . Thus, if we know the concentration of one of the ions of a slightly soluble ionic solid and the value for the solubility product of the solid, then we can calculate the concentration that the other ion must exceed for precipitation to begin. To simplify the calculation, we will assume that precipitation begins when the reaction quotient becomes equal to the solubility product abiding.
Instance ix
Atmospheric precipitation of Calcium Oxalate
Blood will not clot if calcium ions are removed from its plasma. Some blood collection tubes contain salts of the oxalate ion, [latex]\text{C}_2\text{O}_4^{\;\;2-}[/latex], for this purpose (Effigy 4). At sufficiently high concentrations, the calcium and oxalate ions form solid, CaC2O4·H2O (which also contains h2o jump in the solid). The concentration of Ca2+ in a sample of blood serum is 2.two × 10–iii Thousand. What concentration of [latex]\text{C}_2\text{O}_4^{\;\;ii-}[/latex] ion must be established before CaC2O4·H2O begins to precipitate?
Solution
The equilibrium expression is:
[latex]\text{CaC}_2\text{O}_4(s)\;{\rightleftharpoons}\;\text{Ca}^{2+}(aq)\;+\;\text{C}_2\text{O}_4^{\;\;ii-}(aq)[/latex]
For this reaction:
[latex]K_{\text{sp}} = [\text{Ca}^{2+}][\text{C}_2\text{O}_4^{\;\;2-}] = 1.96\;\times\;10^{-8}[/latex]
(see Appendix J)
CaC2O4 does not appear in this expression because it is a solid. Water does not announced because it is the solvent.
Solid CaCtwoO4 does not begin to form until Q equals K sp. Because nosotros know K sp and [Ca2+], we can solve for the concentration of [latex]\text{C}_2\text{O}_4^{\;\;2-}[/latex] that is necessary to produce the showtime trace of solid:
[latex]Q = K_{\text{sp}} = [\text{Ca}^{2+}][\text{C}_2\text{O}_4^{\;\;two-}] = 1.96\;\times\;10^{-8}[/latex]
[latex](ii.2\;\times\;ten^{-3})[\text{C}_2\text{O}_4^{\;\;2-}] = 1.96\;\times\;x^{-8}[/latex]
[latex][\text{C}_2\text{O}_4^{\;\;ii-}] = \frac{1.96\;\times\;10^{-8}}{2.ii\;\times\;x^{-three}} = 8.9\;\times\;ten^{-6}[/latex]
A concentration of [latex][\text{C}_2\text{O}_4^{\;\;2-}][/latex] = 8.9 × 10–6 Yard is necessary to initiate the precipitation of CaC2O4 under these weather condition.
Check Your Learning
If a solution contains 0.0020 mol of [latex]\text{CrO}_4^{\;\;ii-}[/latex] per liter, what concentration of Ag+ ion must exist reached by adding solid AgNO3 before AgtwoCrOfour begins to precipitate? Neglect any increase in volume upon adding the solid silvery nitrate.
It is sometimes useful to know the concentration of an ion that remains in solution subsequently atmospheric precipitation. We can utilise the solubility product for this calculation too: If we know the value of K sp and the concentration of one ion in solution, we can calculate the concentration of the 2nd ion remaining in solution. The calculation is of the same blazon as that in Case 9—calculation of the concentration of a species in an equilibrium mixture from the concentrations of the other species and the equilibrium abiding. However, the concentrations are different; nosotros are computing concentrations subsequently precipitation is complete, rather than at the beginning of precipitation.
Case 10
Concentrations Following Atmospheric precipitation
Vesture washed in water that has a manganese [Mn2+(aq)] concentration exceeding 0.one mg/L (1.8 × 10–half dozen M) may be stained past the manganese upon oxidation, but the corporeality of Mn2+ in the water can be reduced past adding a base. If a person doing laundry wishes to add a buffer to go on the pH loftier enough to precipitate the manganese as the hydroxide, Mn(OH)ii, what pH is required to keep [Mn2+] equal to 1.eight × 10–half-dozen Grand?
Solution
The dissolution of Mn(OH)2 is described by the equation:
[latex]\text{Mn(OH)}_2(s)\;{\rightleftharpoons}\;\text{Mn}^{2+}(aq)\;+\;2\text{OH}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = 2\;\times\;10^{-three}[/latex]
Nosotros need to calculate the concentration of OH– when the concentration of Mn2+ is ane.8 × 10–6 Thou. From that, we calculate the pH. At equilibrium:
[latex]K_{\text{sp}} = [\text{Mn}^{2+}][\text{OH}^{-}]^2[/latex]
or
[latex](1.viii\;\times\;10^{-6})[\text{OH}^{-}]^two = ii\;\times\;ten^{-3}[/latex]
so
[latex][\text{OH}^{-}] = 3.three\;\times\;ten^{-4}\;M[/latex]
Now we calculate the pH from the pOH:
[latex]\text{pOH} = -\text{log}[\text{OH}^{-}] = -\text{log}(3.iii\;\times\;x\;-\;4) = 3.48[/latex]
[latex]\text{pH} = xiv.00\;-\;\text{pOH} = xiv.00\;-\;iii.fourscore = 10.52[/latex]
If the person doing laundry adds a base of operations, such every bit the sodium silicate (NafourSiO4) in some detergents, to the wash water until the pH is raised to x.52, the manganese ion will be reduced to a concentration of ane.8 × 10–6 One thousand; at that concentration or less, the ion will not stain wear.
Bank check Your Learning
The first stride in the preparation of magnesium metallic is the atmospheric precipitation of Mg(OH)2 from body of water water by the addition of Ca(OH)ii. The concentration of Mg2+(aq) in sea h2o is v.37 × 10–2 Yard. Summate the pH at which [Mg2+] is diminished to 1.0 × x–five Yard past the addition of Ca(OH)2.
Due to their light sensitivity, mixtures of silvery halides are used in fiber optics for medical lasers, in photochromic eyeglass lenses (glass lenses that automatically darken when exposed to sunlight), and—earlier the advent of digital photography—in photographic picture show. Even though AgCl (G sp = 1.half-dozen × ten–x), AgBr (Thou sp = 5.0 × x–13), and AgI (K sp = i.5 × 10–16) are each quite insoluble, we cannot prepare a homogeneous solid mixture of them past adding Ag+ to a solution of Cl–, Br–, and I–; substantially all of the AgI will precipitate before any of the other solid halides form because of its smaller value for K sp. However, we can prepare a homogeneous mixture of the solids by slowly adding a solution of Cl–, Br–, and I– to a solution of Ag+.
When two anions form slightly soluble compounds with the same cation, or when two cations form slightly soluble compounds with the aforementioned anion, the less soluble compound (usually, the compound with the smaller Thou sp) generally precipitates offset when we add a precipitating agent to a solution containing both anions (or both cations). When the K sp values of the two compounds differ by two orders of magnitude or more (e.g., 10–ii vs. 10–4), about all of the less soluble compound precipitates before whatsoever of the more soluble 1 does. This is an example of selective precipitation, where a reagent is added to a solution of dissolved ions causing one of the ions to precipitate out before the rest.
The Role of Precipitation in Wastewater Treatment
Solubility equilibria are useful tools in the treatment of wastewater carried out in facilities that may care for the municipal h2o in your urban center or town (Figure five). Specifically, selective precipitation is used to remove contaminants from wastewater earlier it is released back into natural bodies of water. For example, phosphate ions [latex](\text{PO}_4^{\;\;2-})[/latex] are often present in the water discharged from manufacturing facilities. An abundance of phosphate causes excess algae to grow, which impacts the amount of oxygen available for marine life every bit well equally making h2o unsuitable for human consumption.

One common mode to remove phosphates from h2o is by the add-on of calcium hydroxide, known as lime, Ca(OH)two. The lime is converted into calcium carbonate, a strong base of operations, in the water. As the water is fabricated more bones, the calcium ions react with phosphate ions to produce hydroxylapatite, Cafive(PO4)iii(OH), which then precipitates out of the solution:
[latex]v\text{Ca}^{ii+}\;+\;iii\text{PO}_4^{\;\;three-}\;+\;\text{OH}^{-}\;{\leftrightharpoons}\;\text{Ca}_{10}(\text{PO}_4)_6{\cdot}(\text{OH})_2(southward)[/latex]
The precipitate is then removed by filtration and the h2o is brought back to a neutral pH by the add-on of COii in a recarbonation process. Other chemicals tin can as well be used for the removal of phosphates by atmospheric precipitation, including atomic number 26(Three) chloride and aluminum sulfate.
View this site for more than information on how phosphorus is removed from wastewater.
Selective precipitation can also be used in qualitative assay. In this method, reagents are added to an unknown chemical mixture in order to induce precipitation. Sure reagents cause specific ions to precipitate out; therefore, the improver of the reagent can exist used to make up one's mind whether the ion is present in the solution.

View this simulation to study the process of salts dissolving and forming saturated solutions and precipitates for specific compounds, or compounds for which you select the charges on the ions and the K sp
Example 11
Precipitation of Silver Halides
A solution contains 0.0010 mol of KI and 0.10 mol of KCl per liter. AgNO3 is gradually added to this solution. Which forms first, solid AgI or solid AgCl?
Solution
The two equilibria involved are:
[latex]\text{AgCl}(s)\;{\rightleftharpoons}\;\text{Ag}^{+}(aq)\;+\;\text{Cl}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = 1.6\;\times\;10^{-x}[/latex]
[latex]\text{AgI}(south)\;{\rightleftharpoons}\;\text{Ag}^{+}(aq)\;+\;\text{I}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = 1.5\;\times\;ten^{-sixteen}[/latex]
If the solution independent about equal concentrations of Cl– and I–, then the silver salt with the smallest K sp (AgI) would precipitate get-go. The concentrations are not equal, however, and then we should discover the [Ag+] at which AgCl begins to precipitate and the [Ag+] at which AgI begins to precipitate. The table salt that forms at the lower [Ag+] precipitates start.
For AgI: AgI precipitates when Q equals K sp for AgI (one.5 × 10–16). When [I–] = 0.0010 M:
[latex]Q = [\text{Ag}^{+}][\text{I}^{-}] = [\text{Ag}^{+}](0.0010) = 1.five\;\times\;ten^{-16}[/latex]
[latex][\text{Ag}^{+}] = \frac{1.viii\;\times\;10^{-10}}{0.10} = 1.6\;\times\;x^{-ix}[/latex]
AgI begins to precipitate when [Ag+] is ane.v × x–13 M.
For AgCl: AgCl precipitates when Q equals Thousand sp for AgCl (1.6 × 10–10). When [Cl–] = 0.10 M:
[latex]Q_{\text{sp}} = [\text{Ag}^{+}][\text{Cl}^{-}] = [\text{Ag}^{+}](0.10) = 1.six\;\times\;10^{-10}[/latex]
[latex][\text{Ag}^{+}] = \frac{ane.8\;\times\;10^{-ten}}{0.ten} = i.six\;\times\;x^{-nine}\;Yard[/latex]
AgCl begins to precipitate when [Ag+] is ane.six × 10–9 Chiliad.
AgI begins to precipitate at a lower [Ag+] than AgCl, and then AgI begins to precipitate first.
Bank check Your Learning
If silverish nitrate solution is added to a solution which is 0.050 M in both Cl– and Br– ions, at what [Ag+] would atmospheric precipitation begin, and what would be the formula of the precipitate?
Answer:
[Ag+] = one.0 × 10–11 M; AgBr precipitates offset
Common Ion Effect
As we saw when we discussed buffer solutions, the hydronium ion concentration of an aqueous solution of acetic acid decreases when the strong electrolyte sodium acetate, NaCHiiiCOii, is added. We can explain this effect using Le Châtelier's principle. The improver of acetate ions causes the equilibrium to shift to the left, decreasing the concentration of [latex]\text{H}_3\text{O}^{+}[/latex] to compensate for the increased acetate ion concentration. This increases the concentration of CHiiiCO2H:
[latex]\text{CH}_3\text{CO}_2\text{H}\;+\;\text{H}_2\text{O}\;{\rightleftharpoons}\;\text{H}_3\text{O}^{+}\;+\;\text{CH}_3\text{CO}_2^{\;\;-}[/latex]
Because sodium acetate and acerb acid have the acetate ion in common, the influence on the equilibrium is called the common ion effect.
The mutual ion result can also have a direct event on solubility equilibria. Suppose we are looking at the reaction where silvery iodide is dissolved:
[latex]\text{AgI}(s)\;{\rightleftharpoons}\;\text{Ag}^{+}(aq)\;+\;\text{I}^{-}(aq)[/latex]
If nosotros were to add potassium iodide (KI) to this solution, we would exist adding a substance that shares a common ion with silverish iodide. Le Châtelier'due south principle tells us that when a change is fabricated to a organization at equilibrium, the reaction will shift to annul that modify. In this case, there would be an excess of iodide ions, so the reaction would shift toward the left, causing more silvery iodide to precipitate out of solution.

View this simulation to run across how the common ion result work with dissimilar concentrations of salts.
Example 12
Mutual Ion Event
Summate the molar solubility of cadmium sulfide (CdS) in a 0.010-Thousand solution of cadmium bromide (CdBrtwo). The Chiliad sp of CdS is 1.0 × 10–28.
Solution
The first thing you lot should notice is that the cadmium sulfide is dissolved in a solution that contains cadmium ions. We need to utilize an ICE table to gear up this trouble and include the CdBr2 concentration as a correspondent of cadmium ions:
[latex]\text{CdS}(due south)\;{\leftrightharpoons}\;\text{Cd}^{2+}(aq)\;+\;\text{S}^{2-}(aq)[/latex]
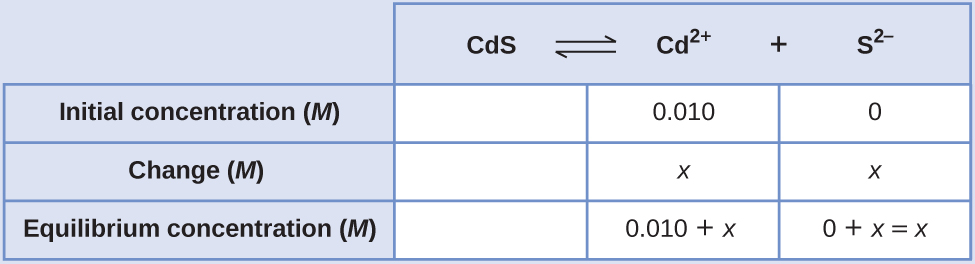
[latex]K_{\text{sp}} = [\text{Cd}^{2+}][\text{South}^{2-}] = 1.0\;\times\;10^{-28}[/latex]
[latex](0.010\;+\;x)(x) = one.0\;\times\;10^{-28}[/latex]
[latex]x^2\;+\;0.010x\;-\;1.0\;\times\;x^{-28} = 0[/latex]
We can solve this equation using the quadratic formula, only we can besides brand an assumption to make this calculation much simpler. Since the K sp value is and so small compared with the cadmium concentration, we tin assume that the modify between the initial concentration and the equilibrium concentration is negligible, so that 0.010 + x ~ 0.010. Going back to our K sp expression, we would now go:
[latex]K_{\text{sp}} = [\text{Cd}^{2+}][\text{South}^{2-}] = 1.0\;\times\;10^{-28}[/latex]
[latex](0.010)(x) = 1.0\;\times\;ten^{-28}[/latex]
[latex]ten = i.0\;\times\;ten^{-26}[/latex]
Therefore, the molar solubility of CdS in this solution is one.0 × 10–26 M.
Check Your Learning
Calculate the tooth solubility of aluminum hydroxide, Al(OH)3, in a 0.015-Thou solution of aluminum nitrate, Al(NO3)iii. The Yard sp of Al(OH)three is 2 × 10–32.
Key Concepts and Summary
The equilibrium constant for an equilibrium involving the atmospheric precipitation or dissolution of a slightly soluble ionic solid is called the solubility product, K sp, of the solid. When nosotros accept a heterogeneous equilibrium involving the slightly soluble solid Chiliad p X q and its ions One thousandm+ and 10n–:
[latex]\text{Thousand}_p\text{10}_q(south)\;{\leftrightharpoons}\;p\text{One thousand}^{\text{g}+}(aq)\;+\;q\text{10}^{\text{n}-}(aq)[/latex]
We write the solubility product expression equally:
[latex]K_{\text{sp}} = [\text{M}^{\text{m}+}]^p[\text{X}^{\text{n}-}]^q[/latex]
The solubility production of a slightly soluble electrolyte can exist calculated from its solubility; conversely, its solubility tin can exist calculated from its M sp, provided the only significant reaction that occurs when the solid dissolves is the formation of its ions.
A slightly soluble electrolyte begins to precipitate when the magnitude of the reaction caliber for the dissolution reaction exceeds the magnitude of the solubility product. Precipitation continues until the reaction quotient equals the solubility production.
A reagent can exist added to a solution of ions to allow one ion to selectively precipitate out of solution. The common ion result tin also play a role in atmospheric precipitation reactions. In the presence of an ion in common with 1 of the ions in the solution, Le Châtelier'southward principle applies and more precipitate comes out of solution so that the molar solubility is reduced.
Key Equations
- [latex]\text{M}_p\text{X}_q(south)\;{\leftrightharpoons}\;p\text{M}^{\text{g}+}(aq)\;+\;q\text{Ten}^{\text{north}-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{M}^{\text{chiliad}+}]^p[\text{X}^{\text{n}-}]^q[/latex]
Chemistry End of Chapter Exercises
- Consummate the changes in concentrations for each of the following reactions:
(a) [latex]\begin{assortment}{lccc} \text{AgI}(s)\;{\longrightarrow}\; & \text{Ag}^{+}(aq) & + & \text{I}^{-}(aq) \\[0.5em] & x & & \rule[0ex]{two.5em}{0.1ex} \end{array}[/latex]
(b) [latex]\begin{assortment}{lccc} \text{CaCO}_3(due south)\;{\longrightarrow} & \text{Ca}^{2+}(aq) & + & \text{CO}_3^{\;\;2-}(aq) \\[0.5em] & \rule[0ex]{2.5em}{0.1ex} & & 10 \end{array}[/latex]
(c) [latex]\brainstorm{array}{lccc} \text{Mg(OH)}_2(s)\;{\longrightarrow} & \text{Mg}^{2+}(aq) & + & two\text{OH}^{-}(aq) \\[0.5em] & x & & \rule[0ex]{2.5em}{0.1ex} \end{array}[/latex]
(d) [latex]\brainstorm{array}{lccc} \text{Mg}_3(\text{PO}_4)_2(s)\;{\longrightarrow} & 3\text{Mg}^{ii+}(aq) & + & 2\text{PO}_4^{\;\;three-}(aq) \\[0.5em] & \rule[0ex]{2.5em}{0.1ex} & & 2x \end{assortment}[/latex]
(e) [latex]\begin{array}{lccccc} \text{Ca}_5(\text{PO}_4)_3\text{OH}(s)\;{\longrightarrow} & 5\text{Ca}^{2+}(aq) & + & 3\text{PO}_4^{\;\;3-}(aq) & + & \text{OH}^{-}(aq) \\[0.5em] & \dominion[0ex]{2.5em}{0.1ex} & & \rule[0ex]{2.5em}{0.1ex} & & x \end{array}[/latex]
- Complete the changes in concentrations for each of the following reactions:
(a) [latex]\begin{array}{lccc} \text{BaSO}_4(s)\;{\longrightarrow} & \text{Ba}^{2+}(aq) & + & \text{SO}_4^{\;\;two-}(aq) \\[0.5em] & ten & & \rule[0ex]{2.5em}{0.1ex} \end{array}[/latex]
(b) [latex]\begin{array}{lccc} \text{Ag}_2\text{So}_4(southward)\;{\longrightarrow} & 2\text{Ag}^{+}(aq) & + & \text{So}_4^{\;\;2-}(aq) \\[0.5em] & \rule[0ex]{ii.5em}{0.1ex} & & ten \end{array}[/latex]
(c) [latex]\begin{array}{lccc} \text{Al(OH)}_3(s)\;{\longrightarrow} & \text{Al}^{3+}(aq) & + & 3\text{OH}^{-}(aq) \\[0.5em] & x & & \rule[0ex]{two.5em}{0.1ex} \finish{array}[/latex]
(d) [latex]\begin{array}{lccccc} \text{Pb(OH)Cl}(s)\;{\longrightarrow} & \text{Atomic number 82}^{two+}(aq) & + & \text{OH}^{-}(aq) & + & \text{Cl}^{-}(aq) \\[0.5em] & \dominion[0ex]{ii.5em}{0.1ex} & & x & & \rule[0ex]{2.5em}{0.1ex} \cease{array}[/latex]
(e) [latex]\begin{array}{lccc} \text{Ca}_3(\text{AsO}_4)_2(s)\;{\longrightarrow} & 3\text{Ca}^{2+}(aq) & + & 2\text{AsO}_4^{\;\;3-}(aq) \\[0.5em] & 3x & & \dominion[0ex]{2.5em}{0.1ex} \end{array}[/latex]
- How do the concentrations of Ag+ and [latex]\text{CrO}_4^{\;\;2-}[/latex] in a saturated solution above 1.0 yard of solid AgiiCrOiv alter when 100 k of solid Ag2CrOfour is added to the organisation? Explain.
- How do the concentrations of Lead2+ and Sii– change when K2Due south is added to a saturated solution of PbS?
- What boosted information do we need to answer the following question: How is the equilibrium of solid silver bromide with a saturated solution of its ions affected when the temperature is raised?
- Which of the following slightly soluble compounds has a solubility greater than that calculated from its solubility product considering of hydrolysis of the anion present: CoSO3, CuI, PbCO3, PbCl2, TliiS, KClOiv?
- Which of the post-obit slightly soluble compounds has a solubility greater than that calculated from its solubility product because of hydrolysis of the anion present: AgCl, BaSO4, CaFii, HgtwoI2, MnCO3, ZnS, PbS?
- Write the ionic equation for dissolution and the solubility production (K sp) expression for each of the following slightly soluble ionic compounds:
(a) PbCl2
(b) Ag2Southward
(c) Sriii(POfour)ii
(d) SrSO4
- Write the ionic equation for the dissolution and the K sp expression for each of the following slightly soluble ionic compounds:
(a) LaF3
(b) CaCO3
(c) Ag2SO4
(d) Pb(OH)2
- The Handbook of Chemical science and Physics gives solubilities of the following compounds in grams per 100 mL of water. Because these compounds are just slightly soluble, assume that the volume does not change on dissolution and calculate the solubility product for each.
(a) BaSiF6, 0.026 g/100 mL (contains [latex]\text{SiF}_6^{\;\;2-}[/latex] ions)
(b) Ce(IO3)4, i.v × 10–2 m/100 mL
(c) Gd2(And then4)3, iii.98 g/100 mL
(d) (NH4)iiPtBr6, 0.59 g/100 mL (contains [latex]\text{PtBr}_6^{\;\;2-}[/latex] ions)
- The Handbook of Chemistry and Physics gives solubilities of the post-obit compounds in grams per 100 mL of water. Because these compounds are simply slightly soluble, assume that the volume does not change on dissolution and calculate the solubility production for each.
(a) BaSeOfour, 0.0118 thou/100 mL
(b) Ba(BrOthree)2·H2O, 0.30 g/100 mL
(c) NH4MgAsOfour·6H2O, 0.038 g/100 mL
(d) Laii(MoO4)iii, 0.00179 chiliad/100 mL
- Utilize solubility products and predict which of the following salts is the most soluble, in terms of moles per liter, in pure h2o: CaF2, Hg2Cl2, PbI2, or Sn(OH)2.
- Assuming that no equilibria other than dissolution are involved, calculate the tooth solubility of each of the following from its solubility product:
(a) KHC4H4O6
(b) PbI2
(c) Agiv[Iron(CN)half-dozen], a table salt containing the [latex]\text{Fe(CN)}_4^{\;\;-}[/latex] ion
(d) Hg2I2
- Assuming that no equilibria other than dissolution are involved, calculate the molar solubility of each of the following from its solubility product:
(a) AgtwoSo4
(b) PbBr2
(c) AgI
(d) CaCtwoO4·H2O
- Assuming that no equilibria other than dissolution are involved, calculate the concentration of all solute species in each of the following solutions of salts in contact with a solution containing a common ion. Show that changes in the initial concentrations of the common ions can be neglected.
(a) AgCl(s) in 0.025 1000 NaCl
(b) CaF2(due south) in 0.00133 M KF
(c) AgiiSOiv(s) in 0.500 L of a solution containing 19.l k of ChiliadiiSOiv
(d) Zn(OH)2(s) in a solution buffered at a pH of 11.45
- Bold that no equilibria other than dissolution are involved, calculate the concentration of all solute species in each of the following solutions of salts in contact with a solution containing a mutual ion. Evidence that changes in the initial concentrations of the common ions tin can exist neglected.
(a) TlCl(s) in i.250 M HCl
(b) PbIii(s) in 0.0355 M CaItwo
(c) Ag2CrO4(s) in 0.225 50 of a solution containing 0.856 g of K2CrOiv
(d) Cd(OH)2(s) in a solution buffered at a pH of 10.995
- Assuming that no equilibria other than dissolution are involved, calculate the concentration of all solute species in each of the following solutions of salts in contact with a solution containing a common ion. Bear witness that information technology is not appropriate to neglect the changes in the initial concentrations of the common ions.
(a) TlCl(s) in 0.025 Yard TlNO3
(b) BaF2(south) in 0.0313 Chiliad KF
(c) MgC2O4 in 2.250 L of a solution containing 8.156 g of Mg(NO3)2
(d) Ca(OH)ii(s) in an unbuffered solution initially with a pH of 12.700
- Explain why the changes in concentrations of the common ions in Chapter xv.1 Chemistry Terminate of Chapter Do 17 can be neglected.
- Explain why the changes in concentrations of the mutual ions in Affiliate 15.i Chemistry End of Chapter Exercise 17 cannot be neglected.
- Summate the solubility of aluminum hydroxide, Al(OH)3, in a solution buffered at pH 11.00.
- Refer to Appendix J for solubility products for calcium salts. Decide which of the calcium salts listed is near soluble in moles per liter and which is most soluble in grams per liter.
- Most barium compounds are very poisonous; however, barium sulfate is frequently administered internally as an aid in the X-ray exam of the lower intestinal tract (Figure 3). This use of BaSOfour is possible because of its low solubility. Calculate the molar solubility of BaSOiv and the mass of barium present in 1.00 L of water saturated with BaSOfour.
- Public Health Service standards for drinking water gear up a maximum of 250 mg/50 (2.60 × x–3 M) of [latex]\text{Then}_4^{\;\;2-}[/latex] because of its cathartic action (information technology is a laxative). Does natural h2o that is saturated with CaSO4 ("gyp" water) every bit a result or passing through soil containing gypsum, CaSO4·2H2O, see these standards? What is [latex]\text{SO}_4^{\;\;ii-}[/latex] in such water?
- Perform the following calculations:
(a) Calculate [Ag+] in a saturated aqueous solution of AgBr.
(b) What will [Ag+] be when plenty KBr has been added to make [Br–] = 0.050 M?
(c) What will [Br–] be when enough AgNOthree has been added to brand [Ag+] = 0.020 M?
- The solubility production of CaSOfour·2H2O is 2.4 × 10–5. What mass of this salt will dissolve in ane.0 Fifty of 0.010 Thou [latex]\text{SO}_4^{\;\;2-}[/latex]?
- Bold that no equilibria other than dissolution are involved, calculate the concentrations of ions in a saturated solution of each of the following (encounter Appendix J for solubility products).
(a) TlCl
(b) BaF2
(c) AgiiCrOiv
(d) CaC2O4·H2O
(east) the mineral anglesite, PbSO4
- Bold that no equilibria other than dissolution are involved, calculate the concentrations of ions in a saturated solution of each of the post-obit (come across Appendix J for solubility products):
(a) AgI
(b) AgtwoAnd sofour
(c) Mn(OH)2
(d) Sr(OH)2·8HtwoO
(eastward) the mineral brucite, Mg(OH)two
- The post-obit concentrations are establish in mixtures of ions in equilibrium with slightly soluble solids. From the concentrations given, calculate K sp for each of the slightly soluble solids indicated:
(a) AgBr: [Ag+] = 5.vii × 10–7 M, [Br–] = 5.seven × ten–7 M
(b) CaCO3: [Ca2+] = five.iii × 10–3 M, [latex][\text{CO}_3^{\;\;ii-}][/latex] = 9.0 × 10–7 M
(c) PbF2: [Pb2+] = 2.1 × 10–3 M, [F–] = 4.ii × ten–3 M
(d) Ag2CrO4: [Ag+] = 5.3 × 10–five M, iii.2 × ten–3 M
(eastward) InF3: [In3+] = 2.3 × x–3 M, [F–] = 7.0 × ten–iii Thousand
- The following concentrations are found in mixtures of ions in equilibrium with slightly soluble solids. From the concentrations given, summate K sp for each of the slightly soluble solids indicated:
(a) TlCl: [Tl+] = ane.21 × 10–2 M, [Cl–] = i.2 × 10–2 M
(b) Ce(IO3)4: [Ce4+] = i.8 × 10–iv Grand, [latex][\text{IO}_3^{\;\;-}][/latex] = 2.6 × 10–13 M
(c) Gd2(SO4)3: [Gd3+] = 0.132 M, [latex][\text{And then}_4^{\;\;2-}][/latex] = 0.198 M
(d) Ag2Thenfour: [Ag+] = 2.xl × 10–2 M, [latex][\text{SO}_4^{\;\;2-}][/latex] = two.05 × ten–2 M
(e) BaSOfour: [Ba2+] = 0.500 M, [latex][\text{SO}_4^{\;\;2-}][/latex] = 2.16 × 10–10 M
- Which of the following compounds precipitates from a solution that has the concentrations indicated? (See Appendix J for K sp values.)
(a) KClO4: [Thousand+] = 0.01 M, [latex][\text{ClO}_4^{\;\;-}][/latex] = 0.01 Thou
(b) Thousand2PtClhalf-dozen: [1000+] = 0.01 Grand, [latex][\text{PtCl}_6^{\;\;2-}][/latex] = 0.01 M
(c) PbI2: [Atomic number 82two+] = 0.003 Thousand, [I–] = ane.three × 10–iii Thou
(d) Ag2S: [Ag+] = i × 10–ten Thou, [S2–] = i × 10–13 1000
- Which of the following compounds precipitates from a solution that has the concentrations indicated? (See Appendix J for K sp values.)
(a) CaCOthree: [Ca2+] = 0.003 Thousand, [latex][\text{CO}_3^{\;\;two-}][/latex] = 0.003 M
(b) Co(OH)2: [Co2+] = 0.01 M, [OH–] = i × 10–seven M
(c) CaHPO4: [Catwo+] = 0.01 M, [latex][\text{HPO}_4^{\;\;2-}][/latex] = 2 × 10–half-dozen Thou
(d) Leadiii(PO4)ii: [Pb2+] = 0.01 One thousand, [latex][\text{PO}_4^{\;\;3-}][/latex] = one × 10–13 Thou
- Calculate the concentration of Tl+ when TlCl just begins to precipitate from a solution that is 0.0250 M in Cl–.
- Calculate the concentration of sulfate ion when BaSOiv just begins to precipitate from a solution that is 0.0758 Thousand in Ba2+.
- Calculate the concentration of Sr2+ when SrF2 starts to precipitate from a solution that is 0.0025 Thousand in F–.
- Summate the concentration of [latex]\text{PO}_4^{\;\;three-}[/latex] when Ag3PO4 starts to precipitate from a solution that is 0.0125 Grand in Ag+.
- Calculate the concentration of F– required to begin atmospheric precipitation of CaF2 in a solution that is 0.010 Thousand in Ca2+.
- Calculate the concentration of Ag+ required to begin atmospheric precipitation of Ag2COthree in a solution that is 2.fifty × ten–6 M in [latex]\text{CO}_3^{\;\;2-}[/latex].
- What [Ag+] is required to reduce [latex][\text{CO}_3^{\;\;2-}][/latex] to 8.2 × 10–4 Thou by precipitation of AgtwoCO3?
- What [F–] is required to reduce [Catwo+] to 1.0 × 10–4 Thou by precipitation of CaFtwo?
- A volume of 0.800 L of a two × 10–four–M Ba(NOiii)2 solution is added to 0.200 L of 5 × ten–4 M Li2Theniv. Does BaSOfour precipitate? Explain your answer.
- Perform these calculations for nickel(II) carbonate. (a) With what book of water must a precipitate containing NiCOiii exist washed to deliquesce 0.100 k of this compound? Assume that the wash water becomes saturated with NiCO3 (K sp = 1.36 × 10–7).
(b) If the NiCOiii were a contaminant in a sample of CoCO3 (1000 sp = 1.0 × 10–12), what mass of CoCO3 would have been lost? Go on in heed that both NiCO3 and CoCO3 dissolve in the aforementioned solution.
- Atomic number 26 concentrations greater than 5.4 × 10–half dozen One thousand in water used for laundry purposes tin cause staining. What [OH–] is required to reduce [Feii+] to this level past precipitation of Iron(OH)2?
- A solution is 0.010 M in both Cu2+ and Cdii+. What per centum of Cd2+ remains in the solution when 99.9% of the Cu2+ has been precipitated as CuS by adding sulfide?
- A solution is 0.15 M in both Leadtwo+ and Ag+. If Cl– is added to this solution, what is [Ag+] when PbClii begins to precipitate?
- What reagent might be used to split the ions in each of the following mixtures, which are 0.ane K with respect to each ion? In some cases information technology may be necessary to command the pH. (Hint: Consider the 1000 sp values given in Appendix J.)
(a) [latex]\text{Hg}_2^{\;\;2+}[/latex] and Cu2+
(b) [latex]\text{SO}_4^{\;\;2-}[/latex] and Cl–
(c) Hg2+ and Co2+
(d) Znii+ and Srtwo+
(east) Ba2+ and Mg2+
(f) [latex]\text{CO}_3^{\;\;two-}[/latex] and OH–
- A solution contains 1.0 × 10–5 mol of KBr and 0.ten mol of KCl per liter. AgNO3 is gradually added to this solution. Which forms first, solid AgBr or solid AgCl?
- A solution contains 1.0 × 10–2 mol of KI and 0.x mol of KCl per liter. AgNO3 is gradually added to this solution. Which forms first, solid AgI or solid AgCl?
- The calcium ions in human blood serum are necessary for coagulation (Effigy 4). Potassium oxalate, ThousandtwoCtwoOiv, is used equally an anticoagulant when a blood sample is drawn for laboratory tests because information technology removes the calcium equally a precipitate of CaC2O4·H2O. It is necessary to remove all but 1.0% of the Catwo+ in serum in guild to forbid coagulation. If normal blood serum with a buffered pH of 7.40 contains 9.five mg of Ca2+ per 100 mL of serum, what mass of MtwoC2Oiv is required to foreclose the coagulation of a 10 mL blood sample that is 55% serum by book? (All volumes are accurate to two pregnant figures. Note that the volume of serum in a 10-mL claret sample is 5.5 mL. Assume that the Ksp value for CaCiiOiv in serum is the same as in water.)
- About 50% of urinary calculi (kidney stones) consist of calcium phosphate, Caiii(POfour)2. The normal mid range calcium content excreted in the urine is 0.10 g of Ca2+ per day. The normal mid range amount of urine passed may be taken every bit 1.iv L per twenty-four hour period. What is the maximum concentration of phosphate ion that urine can incorporate before a calculus begins to form?
- The pH of normal urine is 6.xxx, and the total phosphate concentration [latex]([\text{PO}_4^{\;\;3-}]\;+\;[\text{HPO}_4^{\;\;2-}]\;+\;[\text{H}_2\text{PO}_4^{\;\;-}]\;+\;[\text{H}_3\text{PO}_4])[/latex] is 0.020 Thou. What is the minimum concentration of Ca2+ necessary to induce kidney stone formation? (See Chemistry Terminate of Chapter Practice 49 for additional information.)
- Magnesium metal (a component of alloys used in aircraft and a reducing agent used in the product of uranium, titanium, and other agile metals) is isolated from sea water by the following sequence of reactions:
[latex]\text{Mg}^{2+}(aq)\;+\;\text{Ca(OH)}_2(aq)\;{\longrightarrow}\;\text{Mg(OH)}_2(s)\;+\;\text{Ca}^{2+}(aq)[/latex]
[latex]\text{Mg(OH)}_2(south)\;+\;two\text{HCl}(aq)\;{\longrightarrow}\;\text{MgCl}_2(s)\;+\;2\text{H}_2\text{O}(l)[/latex]
[latex]\text{MgCl}_2(50)\;{\xrightarrow{\text{electrolysis}}}\;\text{Mg}(south)\;+\;\text{Cl}_2(g)[/latex]
Sea water has a density of 1.026 g/cmthree and contains 1272 parts per million of magnesium equally Mg2+(aq) by mass. What mass, in kilograms, of Ca(OH)2 is required to precipitate 99.9% of the magnesium in one.00 × 10three 50 of body of water h2o?
- Hydrogen sulfide is bubbled into a solution that is 0.10 M in both Pb2+ and Fe2+ and 0.xxx Thou in HCl. After the solution has come to equilibrium it is saturated with H2S ([H2S] = 0.x M). What concentrations of Atomic number 822+ and Fe2+ remain in the solution? For a saturated solution of H2Due south we can use the equilibrium:
[latex]\text{H}_2\text{S}(aq)\;+\;2\text{H}_2\text{O}(l)\;{\leftrightharpoons}\;2\text{H}_3\text{O}^{+}(aq)\;+\;\text{S}^{two-}(aq)\;\;\;\;\;\;\;One thousand = 1.0\;\times\;10^{-26}[/latex]
(Hint: The [latex][\text{H}_3\text{O}^{+}][/latex] changes every bit metal sulfides precipitate.)
- Perform the following calculations involving concentrations of iodate ions:
(a) The iodate ion concentration of a saturated solution of La(IOiii)three was found to exist 3.1 × x–3 mol/L. Find the K sp.
(b) Find the concentration of iodate ions in a saturated solution of Cu(IO3)two (M sp = 7.four × x–8).
- Calculate the molar solubility of AgBr in 0.035 M NaBr (K sp = 5 × 10–xiii).
- How many grams of Pb(OH)2 will dissolve in 500 mL of a 0.050-Yard PbCl2 solution (K sp = 1.2 × 10–xv)?
- Use the simulation from the earlier Link to Learning to complete the following practise:. Using 0.01 g CaFtwo, give the Ksp values found in a 0.two-M solution of each of the salts. Discuss why the values change as yous change soluble salts.
- How many grams of Milk of Magnesia, Mg(OH)two (s) (58.3 g/mol), would exist soluble in 200 mL of water. Thousand sp = 7.ane × 10–12. Include the ionic reaction and the expression for G sp in your answer. (K w = ane × 10–xiv = [HiiiO+][OH–])
- Two hypothetical salts, LMii and LQ, have the same tooth solubility in HiiO. If K sp for LM2 is 3.20 × ten–5, what is the K sp value for LQ?
- Which of the following carbonates will course beginning? Which of the following will class last? Explain.
(a) [latex]\text{MgCO}_3\;\;\;\;\;\;\;K_{\text{sp}} = 3.5\;\times\;10^{-viii}[/latex]
(b) [latex]\text{CaCO}_3\;\;\;\;\;\;\;K_{\text{sp}} = 4.2\;\times\;ten^{-7}[/latex]
(c) [latex]\text{SrCO}_3\;\;\;\;\;\;\;K_{\text{sp}} = 3.9\;\times\;x^{-ix}[/latex]
(d) [latex]\text{BaCO}_3\;\;\;\;\;\;\;K_{\text{sp}} = 4.iv\;\times\;10^{-5}[/latex]
(e) [latex]\text{MnCO}_3\;\;\;\;\;\;\;K_{\text{sp}} = 5.one\;\times\;10^{-nine}[/latex]
- How many grams of Zn(CN)2(s) (117.44 g/mol) would be soluble in 100 mL of H2O? Include the balanced reaction and the expression for K sp in your answer. The Thousand sp value for Zn(CN)two(s) is 3.0 × 10–16.
Glossary
- common ion effect
- effect on equilibrium when a substance with an ion in common with the dissolved species is added to the solution; causes a decrease in the solubility of an ionic species, or a decrease in the ionization of a weak acid or base
- tooth solubility
- solubility of a compound expressed in units of moles per liter (mol/L)
- selective precipitation
- process in which ions are separated using differences in their solubility with a given precipitating reagent
- solubility product (Grand sp)
- equilibrium constant for the dissolution of a slightly soluble electrolyte
Solutions
Answers to Chemistry End of Chapter Exercises
1.
(a) [latex]\brainstorm{array}{lccc} \text{AgI}(southward)\;{\rightleftharpoons}\; & \text{Ag}^{+}(aq) & + & \text{I}^{-}(aq) \\[0.5em] & x & & \dominion[-0.25ex]{0.5em}{0.1ex}\hspace{-0.5em}ten \terminate{array}[/latex]
(b) [latex]\begin{array}{lccc} \text{CaCO}_3(southward)\;{\rightleftharpoons} & \text{Ca}^{2+}(aq) & + & \text{CO}_3^{\;\;two-}(aq) \\[0.5em] & \dominion[-0.25ex]{0.5em}{0.1ex}\hspace{-0.5em}x & & ten \end{array}[/latex]
(c) [latex]\brainstorm{array}{lccc} \text{Mg(OH)}_2(southward)\;{\rightleftharpoons} & \text{Mg}^{2+}(aq) & + & two\text{OH}^{-}(aq) \\[0.5em] & x & & \rule[-0.25ex]{1em}{0.1ex}\hspace{-1em}2x \end{array}[/latex]
(d) [latex]\begin{array}{lccc} \text{Mg}_3(\text{PO}_4)_2(due south)\;{\rightleftharpoons} & 3\text{Mg}^{two+}(aq) & + & 2\text{PO}_4^{\;\;iii-}(aq) \\[0.5em] & \dominion[-0.25ex]{1em}{0.1ex}\hspace{-1em}3x & & 2x \end{array}[/latex]
(e) [latex]\brainstorm{array}{lccccc} \text{Ca}_5(\text{PO}_4)_3\text{OH}(s)\;{\rightleftharpoons} & 5\text{Ca}^{ii+}(aq) & + & 3\text{PO}_4^{\;\;3-}(aq) & + & \text{OH}^{-}(aq) \\[0.5em] & \rule[-0.25ex]{1em}{0.1ex}\hspace{-1em}5x & & \rule[-0.25ex]{1em}{0.1ex}\hspace{-1em}3x & & 10 \end{array}[/latex]
three. There is no change. A solid has an activity of 1 whether in that location is a footling or a lot.
5. The solubility of silver bromide at the new temperature must be known. Unremarkably the solubility increases and some of the solid silver bromide will dissolve.
seven. CaF2, MnCO3, and ZnS
9. (a) [latex]\text{LaF}_3(s)\;{\rightleftharpoons}\;\text{La}^{3+}(aq)\;+\;3\text{F}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{La}^{3+}][\text{F}^{-}]^3[/latex];
(b) [latex]\text{CaCO}_3(s)\;{\rightleftharpoons}\;\text{Ca}^{2+}(aq)\;+\;\text{CO}_3^{\;\;ii-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Ca}^{ii+}][\text{CO}_3^{\;\;2-}][/latex];
(c) [latex]\text{Ag}_2\text{SO}_4(southward)\;{\rightleftharpoons}\;2\text{Ag}^{+}(aq)\;+\;\text{SO}_4^{\;\;2-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Ag}^{+}]^ii[\text{Then}_4^{\;\;2-}][/latex];
(d) [latex]\text{Pb(OH)}_2(south)\;{\rightleftharpoons}\;\text{Pb}^{2+}(aq)\;+\;2\text{OH}^{-}(aq)\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Pb}^{2+}][\text{OH}^{-}]^2[/latex]
11. (a)1.77 × ten–vii; (b) one.six × 10–half dozen; (c) 2.two × 10–9; (d) vii.91 × 10–22
13. (a) two × ten–two M; (b) ane.5 × 10–3 M; (c) two.27 × ten–9 One thousand; (d) 2.2 × 10–ten M
15. (a) vi.iv × 10−9 M = [Ag+], [Cl−] = 0.025 Grand
Cheque: [latex]\frac{half dozen.4\;\times\;10^{-9}\;M}{0.025\;M}\;\times\;100\% = 2.half-dozen\;\times\;10^{-five}\;%[/latex],an insignificant change;
(b) 2.ii × 10−v M = [Ca2+], [F−] = 0.0013 K
Check: [latex]\frac{2.26\;\times\;ten^{-five}\;Yard}{0.00133\;M}\;\times\;100\% = 1.70\%[/latex]. This value is less than 5% and tin be ignored.
(c) 0.2238 M = [latex][\text{Then}_4^{\;\;2-}][/latex]; [Ag+] = 7.4 × x–3 Yard
Check: [latex]\frac{3.7\;\times\;x^{-3}}{0.2238}\;\times\;100\% = 1.64\;\times\;10^{-2}\%[/latex]; the condition is satisfied.
(d) [OH–] = two.viii × 10–three M; 5.vii × 10−12 Grand = [Zn2+]
Check: [latex]\frac{v.7\;\times\;10^{-12}}{2.8\;\times\;10^{-iii}}\;\times\;100\% = ii.0\;\times\;10^{-seven}\%[/latex]; x is less than 5% of [OH–] and is, therefore, negligible.
17. (a) [Cl–] = 7.half dozen × ten−iii M
Bank check: [latex]\frac{7.6\;\times\;10^{-3}}{0.025}\;\times\;100\% = thirty\%[/latex]
This value is also large to drib x. Therefore solve past using the quadratic equation:
[Ti+] = 3.1 × 10–2 M
[Cl–] = 6.one × 10–three
(b) [Ba2+] = 7.7 × 10–4 K
Check: [latex]\frac{7.7\;\times\;10^{-4}}{0.0313}\;\times\;100\% = 2.4\%[/latex]
Therefore, the status is satisfied.
[Batwo+] = seven.vii × 10–4 Thousand
[F–] = 0.0321 Thou;
(c) Mg(NO3)2 = 0.02444 M
[latex][\text{C}_2\text{O}_4^{\;\;ii-}] = ii.9\;\times\;x^{-five}[/latex]
Check: [latex]\frac{2.9\;\times\;ten^{-5}}{0.02444}\;\times\;100\% = 0.12\%[/latex]
The status is satisfied; the to a higher place value is less than 5%.
[latex][\text{C}_2\text{O}_4^{\;\;2-}] = 2.nine\;\times\;10^{-5}\;1000[/latex]
[Mg2+] = 0.0244 M
(d) [OH–] = 0.0501 Yard
[Ca2+] = 3.fifteen × 10–3
Check: [latex]\frac{3.15\;\times\;10^{-3}}{0.050}\;\times\;100\% = vi.28\%[/latex]
This value is greater than v%, so a more than exact method, such every bit successive approximations, must be used.
[Caii+] = 2.eight × 10–3 M
[OH–] = 0.053 × ten–ii Thousand
19. The changes in concentration are greater than v% and thus exceed the maximum value for disregarding the change.
21. CaSO4∙2H2O is the most soluble Ca salt in mol/50, and information technology is too the nigh soluble Ca salt in g/L.
23. 4.eight × 10–3 One thousand = [latex][\text{SO}_4^{\;\;ii-}][/latex] = [Ca2+]; Since this concentration is higher than 2.60 × 10–3 M, "gyp" water does not see the standards.
25. Mass (CaSO4·2H2O) = 0.72 grand/50
27. (a) [Ag+] = [I–] = 1.three × ten–v M; (b) [Ag+] = ii.88 × ten–2 Yard, [latex][\text{And so}_4^{\;\;2-}][/latex] = i.44 × x–2 Yard; (c) [Mn2+] = 3.7 × 10–v One thousand, [OH–] = seven.4 × 10–v M; (d) [Srtwo+] = 4.3 × 10–ii M, [OH–] = 8.6 × x–2 M; (e) [Mg2+] = 1.3 × 10–4 M, [OH–] = 2.6 × 10–iv M.
29. (a) ii.0 × 10–iv; (b) 5.1 × 10–17; (c) ane.35 × 10–4; (d) 1.18 × 10–v; (e) 1.08 × ten–10
31. (a) CaCO3 does precipitate.
(b) The compound does not precipitate.
(c) The compound does not precipitate.
(d) The chemical compound precipitates.
33. 3.03 × 10−7 G
35. ix.two × 10−13 M
37. [Ag+] = 1.eight × 10–3 M
39. 6.iii × 10–four
41. (a) 2.25 L; (b) 7.ii × 10–7 one thousand
43. 100% of information technology is dissolved
45. (a) [latex]\text{Hg}_2^{\;\;2+}[/latex] and Cu2+: Add together [latex]\text{Then}_4^{\;\;2-}[/latex].
(b) [latex]\text{SO}_4^{\;\;2-}[/latex] and Cl–: Add Ba2+.
(c) Hgtwo+ and Co2+: Add S2–.
(d) Zn2+ and Srii+: Add OH– until [OH–] = 0.050 M.
(due east) Ba2+ and Mgtwo+: Add together [latex]\text{SO}_4^{\;\;2-}[/latex].
(f) [latex]\text{CO}_3^{\;\;2-}[/latex] and OH–: Add Baii+.
47. AgI will precipitate showtime.
49. 1.five × 10−12 One thousand
51. three.99 kg
53. (a) 3.1 × 10–xi; (b) [Cu2+] = 2.6 × 10–iii; [latex][\text{IO}_3^{\;\;-}][/latex] = 5.3 × 10–3
55. 1.8 × x–5 yard Atomic number 82(OH)2
57. [latex]\text{Mg(OH)}_2(s)\;{\rightleftharpoons}\;\text{Mg}^{ii+}\;+\;2\text{OH}^{-}\;\;\;\;\;\;\;K_{\text{sp}} = [\text{Mg}^{two+}][\text{OH}^{-}]^2[/latex]
one.23 × 10−3 m Mg(OH)two
59. MnCO3 will course showtime, since it has the smallest K sp value information technology is the least soluble. MnCOthree will be the terminal to precipitate, it has the largest K sp value.
gordonberaing1955.blogspot.com
Source: https://opentextbc.ca/chemistry/chapter/15-1-precipitation-and-dissolution/
0 Response to "What Will the Concentration of Cd2+ at the Moment Before Fe2+ Begins to Precipitate?"
Post a Comment